Physical Science Vocabulary, notes & Review SECTION
Physical Science Vocabulary
Quiz 1
1. Matter-anything that has mass and takes up space.
2. Mass-The amount of matter in an object
3. Volume -Amount of space an object takes up (V=LxWxH)
4. Property - Characteristics of a substance
5. Characteristic - A feature that helps identify an object
6. Density - A measure of how tightly packed matter
7. element – basic building block of matter, pure substance that cannot be broken down.
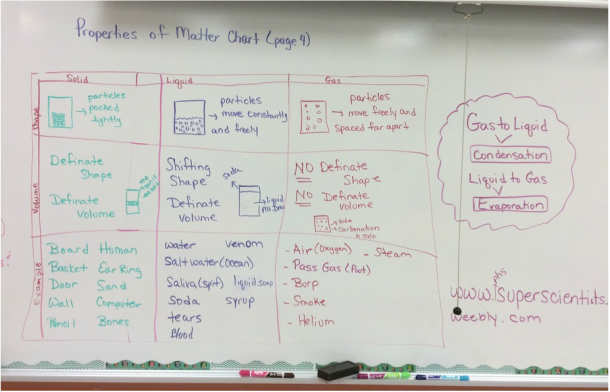
8. States of Matter – the forms that matter can take – solid, liquid, gas
9. solid – a state of matter in which a substance has a definite shape and a definite volume. The molecules in a solid move very slowly.
10. liquid – a state of matter in which a substance has a definite volume but takes the shape of its container. The molecules in a liquid move faster than those in a solid.
11. gas – the state of matter in which the substance takes both the shape and the volume of its container. The molecules in a gas move very quickly and freely.

Quiz 2
12. physical property – a property that can be observed, measured, or changed without changing the substance itself
13. physical change – a change from one form to another form without turning into a new substance
14. chemical change – a change that produces a new substance
15. chemical reaction-a chemical change of original substances into one or more new substances
16. reactant-one of the original substance before a chemical reaction takes place
Quiz 3
17. heat energy – the ways in which heat can travel from one object or substance to another (also called thermal energy)
18. Temperature – the average speed of the particles in a substance
19. Heat transfer – the ways in which heat can travel from one object or substance to another
20. Kinetic energy-the energy of a moving object (sliding down the slide)
21. Potential energy-the energy stored in an object or material (sitting at the top of a slide)
22. Conduction-the movement of heat between objects that TOUCH each other
23. Convection – the movement of heat energy through LIQUIDS and GASES in CURRENTS
24. Radiation – the movement of heat energy through SPACE AS WAVES
25. Conductor - a material that allows heat energy or electricity to pass through it easily (example - metal)
26. Insulator - a material that does not let heat energy or electricity pass through it easily (example - wood)
Examples:
Conductor Insulators
1. metal 1. wood
2. Silver 2. plastic
3. Gold 3. glass
4. Copper 4. rubber
5. Tin 5. wool
6. Steel 6. Leather
12. physical property – a property that can be observed, measured, or changed without changing the substance itself
13. physical change – a change from one form to another form without turning into a new substance
14. chemical change – a change that produces a new substance
15. chemical reaction-a chemical change of original substances into one or more new substances
16. reactant-one of the original substance before a chemical reaction takes place
Quiz 3
17. heat energy – the ways in which heat can travel from one object or substance to another (also called thermal energy)
18. Temperature – the average speed of the particles in a substance
19. Heat transfer – the ways in which heat can travel from one object or substance to another
20. Kinetic energy-the energy of a moving object (sliding down the slide)
21. Potential energy-the energy stored in an object or material (sitting at the top of a slide)
22. Conduction-the movement of heat between objects that TOUCH each other
23. Convection – the movement of heat energy through LIQUIDS and GASES in CURRENTS
24. Radiation – the movement of heat energy through SPACE AS WAVES
25. Conductor - a material that allows heat energy or electricity to pass through it easily (example - metal)
26. Insulator - a material that does not let heat energy or electricity pass through it easily (example - wood)
Examples:
Conductor Insulators
1. metal 1. wood
2. Silver 2. plastic
3. Gold 3. glass
4. Copper 4. rubber
5. Tin 5. wool
6. Steel 6. Leather
Prior Knowledge for Students on PHYSICAL SCIENCE
Created and Copied From LENOIR COUNTY PUBLIC SCHOOL
All matter has mass and volume. Mass is a measure of the amount of matter in an object and is measured in grams. Mass is conserved, so the total mass of an object is always equal to the sum of its parts. However, in practice, students may find small differences when they measure the mass of an object and its parts because of measurement errors and instrument precision. Mass is measured with a balance.
Weight is a measure of the pull of gravity on the mass of an object. It depends both on the mass of an object and the force of gravity. More-massive objects always weigh more than less-massive objects if they are weighed in the same place. However, an object on the moon may weigh less than an object that has less mass on Earth because the force of gravity is less on the moon. As described by the universal law of gravity, the force of gravity depends directly on the mass of the objects and inversely to the square of the distance between them. Consequently, astronauts orbiting Earth have very little weight and appear weightless because they are far from Earth. Weight is measured in Newtons in the metric system or pounds in the U.S, system. Scales are used to measure weight. Balances are used to measure mass.
In a chemical change, the chemical composition of the substances involved is changed. The result is a new substance that has new chemical and physical properties. Physical changes, on the other hand, only change physical properties of the material. The identity of the substance, and thus its chemical properties, does not change as the result of a physical change. For example, cutting wood into small pieces is a physical change that changes the size and the shape of the wood, but the small pieces of wood still burn. However, burning the wood is a chemical change that forms substances that are not flammable.
Many chemical changes are difficult to reverse and cannot be reversed by physical means, but it is not accurate to say that they cannot be reversed. For example, electricity can be used to separate water molecules into oxygen and hydrogen atoms.
Chemical reactions may be fast or slow. Rusting is a chemical reaction between iron and oxygen that occurs relatively slowly, whereas a very fast chemical reaction causes fireworks to explode. Even though rusting is a slow reaction, it does give off some heat energy. A reaction that produces heat energy is called an exothermic reaction. The combustion of the fuel in fireworks is extremely exothermic and gives off large amounts of energy in the form heat and light. An endothermic reaction uses more heat energy than it produces, causing the temperature of the product to decrease. The addition of heat is needed to start some reactions. A simple example of this is the addition of heat to cook food. Light energy can also initiate certain reactions, which is why some products are packaged in containers that block light.
Heat energy is a form of energy that affects the way particles in a substance move. Heat energy moves from a hot object to a cooler object, until they are both the same temperature. Heat travels better through materials that are conductors, like metals. Heat travels poorly though materials that are insulators, like some foams and plastics. When heat travels, it can travel between substances that are touching. This is called conduction. Heat can also travel in the form of a wave. This is called thermal radiation. Thermal radiation occurs when objects emit heat energy as a wave. A third means of heat energy transport involves heat transfer through fluids by convection. Convection occurs when hot portions of fluid move away from the heat source, allowing heat to travel through the entire fluid. For example, convection space heaters transfer heat to air, which is the fluid that transports heat to warm your body.
Heat energy, or thermal energy, is the energy of moving particles. Particles that are hotter move faster, and particles that are colder move slower. There is no such thing as cold energy, only the absence of heat energy.
When energy is used to make particles move faster, heat energy is released. This happens when light energy strikes a surface and is absorbed; particles on the surface move faster, and are therefore hotter. The same thing occurs when two objects rub against each other; the friction transfers kinetic energy to the particles, which absorb the energy, move faster, and are therefore hotter. When a chemical reaction occurs, sometimes chemical energy is released, causing the particles in the reaction to move faster. This means the particles have more heat energy.
Heat energy is transferred when two materials whose particles have different average heat energies come into contact with each other. Heat energy flows from the material with more average heat energy to the material with less average heat energy – from hot to cold. This is why objects that are warm release heat energy into the surrounding environment. For example, if a space heater is surrounded by cool air, heat energy will flow from the hot heater to the cool air around it, warming the air particles. Some materials are resistant to the transfer of heat energy. These materials are called insulators.
Created and Copied From LENOIR COUNTY PUBLIC SCHOOL
All matter has mass and volume. Mass is a measure of the amount of matter in an object and is measured in grams. Mass is conserved, so the total mass of an object is always equal to the sum of its parts. However, in practice, students may find small differences when they measure the mass of an object and its parts because of measurement errors and instrument precision. Mass is measured with a balance.
Weight is a measure of the pull of gravity on the mass of an object. It depends both on the mass of an object and the force of gravity. More-massive objects always weigh more than less-massive objects if they are weighed in the same place. However, an object on the moon may weigh less than an object that has less mass on Earth because the force of gravity is less on the moon. As described by the universal law of gravity, the force of gravity depends directly on the mass of the objects and inversely to the square of the distance between them. Consequently, astronauts orbiting Earth have very little weight and appear weightless because they are far from Earth. Weight is measured in Newtons in the metric system or pounds in the U.S, system. Scales are used to measure weight. Balances are used to measure mass.
In a chemical change, the chemical composition of the substances involved is changed. The result is a new substance that has new chemical and physical properties. Physical changes, on the other hand, only change physical properties of the material. The identity of the substance, and thus its chemical properties, does not change as the result of a physical change. For example, cutting wood into small pieces is a physical change that changes the size and the shape of the wood, but the small pieces of wood still burn. However, burning the wood is a chemical change that forms substances that are not flammable.
Many chemical changes are difficult to reverse and cannot be reversed by physical means, but it is not accurate to say that they cannot be reversed. For example, electricity can be used to separate water molecules into oxygen and hydrogen atoms.
Chemical reactions may be fast or slow. Rusting is a chemical reaction between iron and oxygen that occurs relatively slowly, whereas a very fast chemical reaction causes fireworks to explode. Even though rusting is a slow reaction, it does give off some heat energy. A reaction that produces heat energy is called an exothermic reaction. The combustion of the fuel in fireworks is extremely exothermic and gives off large amounts of energy in the form heat and light. An endothermic reaction uses more heat energy than it produces, causing the temperature of the product to decrease. The addition of heat is needed to start some reactions. A simple example of this is the addition of heat to cook food. Light energy can also initiate certain reactions, which is why some products are packaged in containers that block light.
Heat energy is a form of energy that affects the way particles in a substance move. Heat energy moves from a hot object to a cooler object, until they are both the same temperature. Heat travels better through materials that are conductors, like metals. Heat travels poorly though materials that are insulators, like some foams and plastics. When heat travels, it can travel between substances that are touching. This is called conduction. Heat can also travel in the form of a wave. This is called thermal radiation. Thermal radiation occurs when objects emit heat energy as a wave. A third means of heat energy transport involves heat transfer through fluids by convection. Convection occurs when hot portions of fluid move away from the heat source, allowing heat to travel through the entire fluid. For example, convection space heaters transfer heat to air, which is the fluid that transports heat to warm your body.
Heat energy, or thermal energy, is the energy of moving particles. Particles that are hotter move faster, and particles that are colder move slower. There is no such thing as cold energy, only the absence of heat energy.
When energy is used to make particles move faster, heat energy is released. This happens when light energy strikes a surface and is absorbed; particles on the surface move faster, and are therefore hotter. The same thing occurs when two objects rub against each other; the friction transfers kinetic energy to the particles, which absorb the energy, move faster, and are therefore hotter. When a chemical reaction occurs, sometimes chemical energy is released, causing the particles in the reaction to move faster. This means the particles have more heat energy.
Heat energy is transferred when two materials whose particles have different average heat energies come into contact with each other. Heat energy flows from the material with more average heat energy to the material with less average heat energy – from hot to cold. This is why objects that are warm release heat energy into the surrounding environment. For example, if a space heater is surrounded by cool air, heat energy will flow from the hot heater to the cool air around it, warming the air particles. Some materials are resistant to the transfer of heat energy. These materials are called insulators.